CAUSTIC
Caustic soda (sodium hydroxide) is the most widely used and available alkaline material. Most caustic soda is produced as a co-product in the production of chlorine by the electrolysis of sodium chloride. The caustic solution is concentrated in a series of evaporators. The presence of chlorides and sodium chlorate also add to the corrosiveness of the process.
Caustic soda is generally supplied in concentrations of 50%, 73% or anhydrous. The largest consumer of caustic soda is the chemical process industry, but it is also widely used in the pulp and paper industry and in the aluminum industry.
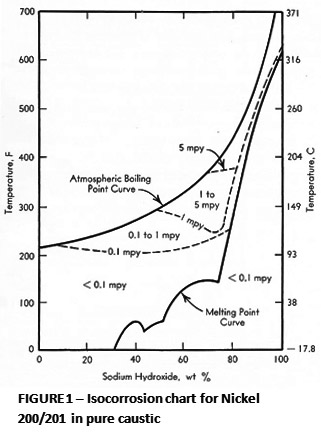
Depending on concentration, temperature and impurities, a wide range of alloys may be considered for use in caustic service. Increasing nickel content improves the resistance of stainless and nickel alloys to caustic. Commercially pure nickel alloys 200 and 201 are the most resistant to caustic corrosion and are widely used. Pure nickel alloys also appear to be immune to caustic embrittlement. Figure 1 illustrates the general corrosion resistance of these two nickel alloys. Nickel chromium alloys such as Alloy 600 also exhibit very useful resistance. Alloy 600 can suffer from caustic cracking under severe conditions, such as 50% caustic at temperatures above 300° C, in air. Under oxygen free conditions, no cracking occurs.
The resistance of copper and copper nickel alloys is fundamentally a function of how much nickel they contain. The copper base alloys do offer resistance at moderate temperatures in the 25% to 73% range, but are generally not used because copper contamination of the product is undesirable. In addition, chlorate and hypochlorite contamination is detrimental to their performance. The nickel copper alloys 400 and K500 are resistant to caustic environments, approaching the resistance of Nickel 200. Under very high stress and elevated temperatures these materials can suffer from caustic cracking.
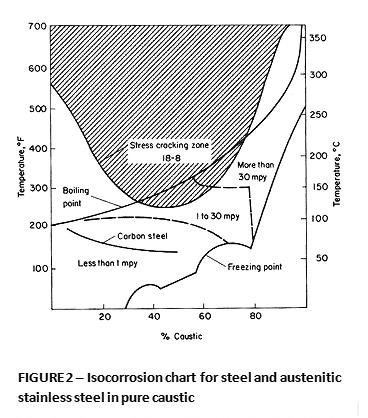
Cast iron or steel are adequate for low temperature use, particularly if iron contamination of the product is not an issue. At elevated temperature, steel alloys can be susceptible to caustic embrittlement and excessive corrosion. Stress relief of fabrications and welds can extend the range of useful service conditions. Stainless steels have useful general corrosion resistance in a broad range of conditions, but are also sensitive to caustic stress corrosion cracking (CSCC) at temperatures above about 200° F. Figure 2 illustrates the resistance of 300 series stainless steels in pure caustic. There is little difference in the general corrosion resistance of alloys like 304, 316 and 310 provided they are in the solution annealed condition.
More highly alloyed stainless steels, like the 6% molybdenum AL-6XN® Alloy also offer useful resistance to caustic environments. Testing of AL-6XN alloy in simulated caustic evaporator solutions containing 43% sodium hydroxide in the presence of sodium chlorate and sodium chloride at up to 190° C resulted in rates just slightly above those of Alloy 200. No stress cracking was observed. Other higher nickel stainless alloys like Alloy 20, 800 and 825 also resist caustic corrosion and caustic cracking better than the 300 series stainless steels. These alloys can be candidates for service in caustic exposure when corrosion from cooling waters is a concern.
Duplex stainless alloys also show resistance to caustic and caustic cracking. Alloy 2205 is useful up to 60% concentration at 90° C with no evidence of caustic cracking. Super Duplex alloys like ZERON® 100 will offer slightly greater resistance than 2205, which benefit from higher chromium and nickel levels. Titanium alloys can be resistant up to about 40% concentration and below 80° C. Generally, titanium is less resistant than alloy 200. Zirconium alloys offer resistance up to 50% and 60° C. Tantalum is not resistant to caustic solutions.